Medical Device Research Business
#Core business ·2025-05-09 14:06:01
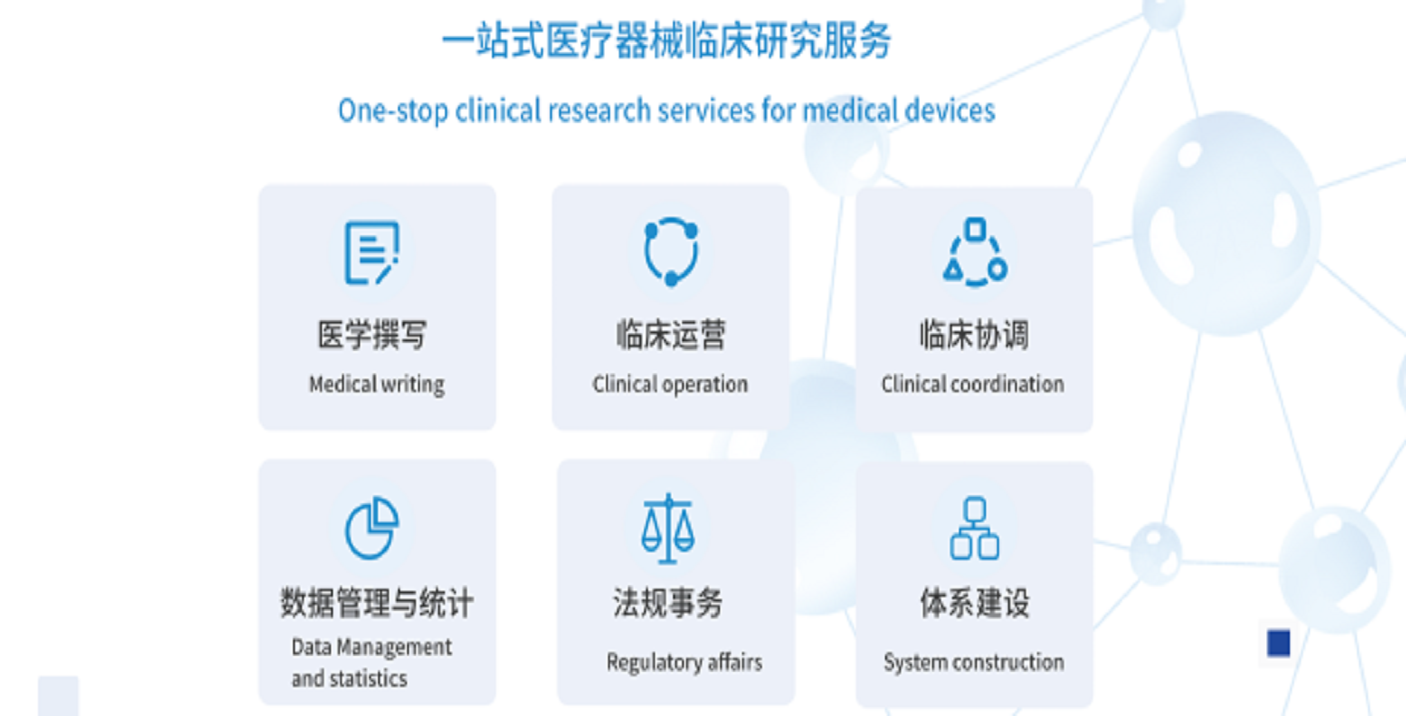
Under Totrial, with Feiwei Pharmaceutical (Shanghai) Technology Co., Ltd. as the main body, it provides one-stop medical device clinical research services for device customers, integrating medical writing, clinical operations, clinical coordination, data management and statistics, regulatory affairs, and system construction. Focusing on clinical research and registration application in the field of medical devices for nearly a decade, it has established in-depth cooperative relationships with the translational medicine platform of Capital Medical University, the translational medicine platform of Shanghai Jiao Tong University, and various parks in the Beijing-Tianjin-Hebei region and the Yangtze River Delta, creating an integrated platform for medical-engineering transformation for medical device enterprises. Over the past decade, the fields of medical devices involved have covered neuroregulation implantable and non-implantable products, auxiliary diagnostic software products, energy-based treatment equipment, and new material (orthopedic and medical aesthetics) implantable products. It has successfully approved products for many domestic listed medical device enterprises and unicorn companies, and several products have obtained the first marketing license in the same field.